My life as an organ-on-a-chip
Diary notes from the perspective of an organ-on-chip
Monday
Finally, it’s Monday morning, which means that I get some new medium (food) and that I get to play the main role in some exiting experiments that are planned this week. Let me first introduce myself: I’m an organ on a chip, specifically a heart-on-a-chip. Last week, I was created from human induced pluripotent stem cell derived cardiomyocytes (heart cells), fibroblasts (constructive tissue cells) and endothelial cells (blood vessel cells) mixed in a collagen gel (extracellular matrix). This mix was seeded on a chip platform with two flexible pillars, where the cell-gel mix compacts and self-organizes in a way that it forms a heart tissue that anchors and starts pulling the pillars when contraction start. That’s how a heart-on-chip is made! Luckily, I started spontaneous beating three days after formation. My creator, Laura Windt, who I see looking up into the microscope, came checking up on me already early in the morning and gave me some food, told me I look beautiful, took photos of me through the microscope and left me alone again, as she ran off to the Monday morning stem cell meeting, where every week someone from the lab group gives a nice overview on their stem cell related work. After the meeting, she had finally time to measure my contraction rate. It was a bit slow, but with a pacing stimulation (just like a pacemaker) it increased to 1.2 Hz (=72 beats per minute), which is more physiologically relevant. During this experiment the pacing stimulation was upregulated to 3 Hz, wherefore I was contracting with 180 beats per minute!
Tuesday
This day I have a well-deserved easy and quiet day after yesterday’s workout. I am chilling in the warm and cozy incubator where it’s always 37C, 5% CO2 and a perfect humidity. However, I know that I am the protagonist in a meeting this morning as the team want to know exactly how I have grown over the past week and the condition of my cellular organization. Far away, I can hear the brainstorm session on immunofluorescence staining and clearing prior to imaging with a confocal microscope. And later they talk about histology sections, seems painful, I would prefer an MRI. It feels like they are always trying to find a way to visualize me in 3D, seems very challenging (people working with 3D tissues can probably relate). They decided to already fix some heart tissues tomorrow for histology sections and staining, what a quick and efficient process! Then, a lot of researchers came in the lab to take some pictures for the website while “working in the lab”. Then, there was a floor meeting and I heard work that was being done on the computer.
Wednesday
Today, some of my siblings, my neighbour heart-on-chips, were removed and fixed. Luckily, I still get to live! The tissues on the chips were taken out, transferred to another well plate, washed and a fixation solvent was added. However, the chips started floating instead of staying on the bottom of the well. Laura says that there is always something unexpected happening when you work with organs-on-chips! Luckily there were colleagues present that gave advice to glue the chips to the bottom of the well with a substance called High Vacuum Grease, after which colleague Jeroen started singing Grease Lightning! For the next experiment, a substance with fluorescent dyes were added to my medium. After a while, most of my cells started shining in a beautiful blue color and just some of them were green, which means that I am mostly composed of living cells and almost none are dying. A lot of pictures were taken under the microscope and a 3D reconstruction was made of the blue lights inside of me, it reminds me of a night sky!
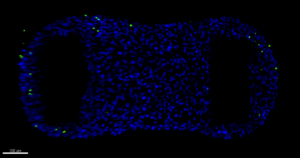
“Selfie of heart-on-a-chip”
Thursday
As usual, the day begins with Laura checking on me, feeding and again a photoshoot. When I look next to me, I see that there are not many siblings left in the wells next to me. Is this how the fittest heart tissues survive? Little did I know that the real test was yet to come. I was moved to the microscope and some pacing electrodes were placed next to me, all seemed pretty similar as the experiment on Monday. But then they gave me some new medium, again? Whooooaaaah, I started beating faster and faster. There must have been some kind of compound in my medium! On the medium flask it says: Isoprenaline. The medium was replaced again and now my contraction rate was so high that it gets very exhausting. Finally, the medium was replaced with normal medium without that scary compound and I had time to recover in the incubator.
Friday
Today Laura checks the quality of the heart cells that were used to make me and my siblings. That was done by FACS, where you can check for the purity of the cells. I already know that I am of good quality, as I look very good, contract spontaneously and respond very well to pacing. However, they always want to know for sure. After confirmation I am definitely of good quality we continue with the next experiments. My contraction rate is checked again, it has increased a bit, might be because of the stress of yesterday. Luckily, I am able to follow all the pacing stimulations. But then suddenly Laura leaves me alone and opens her laptop. She starts talking about a review they are working on, I hear her talking about other organ-on-chip types, which makes me very curious! After finishing the experiments on contraction and viability, I have been put back in the incubator. Luckily, I know that she will be back in the lab on Sunday, as we have these experiments together every other day.
 Laura Windt, NOCI PhD-student at LUMC
Laura Windt, NOCI PhD-student at LUMC
Heart-on-Chip, Anatomy and Embryology lab of Prof. Dr. Christine Mummery
