If you would have told me 15 years ago that I would spend my PhD transforming cells that are found in urine towards intestinal epithelial cells and into a miniature intestine on-chip, I would not have believed it. Even today, it still sounds a bit like science fiction. Nevertheless, it accurately depicts what I have been working on in the last 4,5 years of my PhD project.
Together with my fellow PhD colleague Joram Mooiweer and under supervision of Sebo Withoff and Iris Jonkers, we were able to generate ‘induced pluripotent stem cells’ (iPSCs) from mature differentiated cells, derived from urine or blood from selected individuals. These iPSCs are somatic cells that are genetically reprogrammed to acquire a stem cell phenotype and thus have the ability to transform into any cell type of the human body when exposed to the right growth factors. We made use of this characteristic and guided these cells towards intestinal epithelial organoids by following protocols published by the group of James Wells. Subsequently, we introduced these iPSC-derived intestinal epithelial cells in the upper channel of an Emulate Intestine-Chip system and exposed the cells to flow. After a few days, the tissue developed into an extensive network of 3D folds that resembles the villi we have in the small intestine.
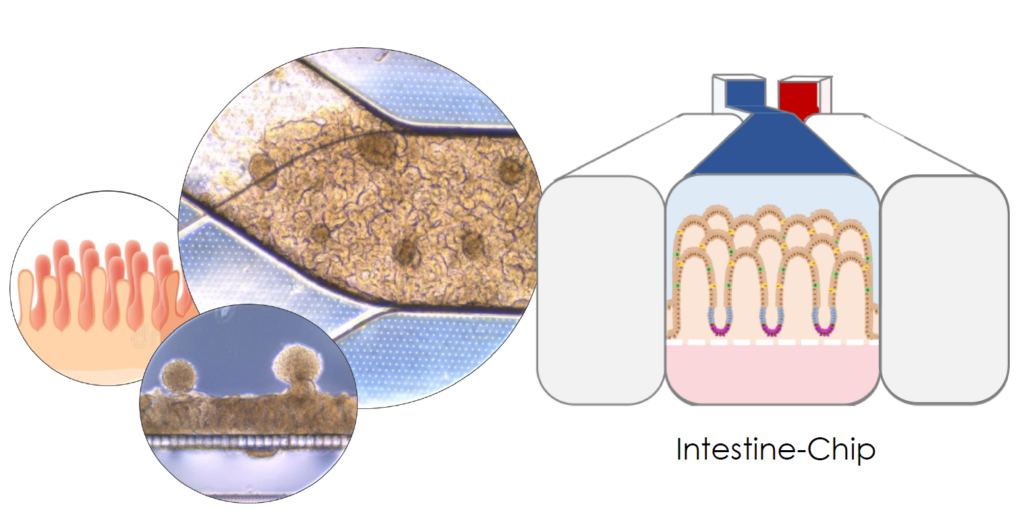
Intestine-Chip containing intestinal epithelial tissue that developed into fold-like structures. This figure was adapted from a publication by Renée Moerkens, Joram Mooiweer et al.: https://doi.org/10.1177/2050640619836057
Even though the tissue looked quite similar to the human intestine in morphology, we wanted to further assess whether the cell type composition reflected the diverse epithelial subpopulations that reside in our intestine. The intestinal epithelial barrier is composed of different subtypes that each have different roles: for example, hormone production, digestion of nutrients or the regeneration or protection of the intestinal barrier. By tweaking the medium composition and exposing the cells to an environment that resembles the growth factor gradients that are present along the intestinal villi, we could steer the intestinal epithelial subtype composition from stem cell-enriched tissue to a more differentiated and functional tissue, that better reflects the intestinal epithelial composition in the human intestine. To find the right media conditions, we received a lot of help from our NOCI collaborators in the group of Hans Clevers at the Hubrecht Institute, who studied these processes extensively in adult stem cell derived-intestinal organoids.
Last July I got the opportunity to present this work during the annual European Organ-on-Chip Society (EUROoCS) conference in Grenoble. Being able to discuss this data with many enthusiastic researchers in the field was a great way to obtain relevant new insights. And as cherry on top of the cake, I was awarded the Best Oral Presentation Award, which was a very special recognition for the years of work during my PhD.

Part of the NOCI consortium during the EUROoCS 2022 conference in Grenoble.
These past years have taught me that, although the Organ-on-Chip systems may look simplistic in design, setting-up these systems requires a great deal of technological as well as biological knowledge. This is what has made the Netherlands Organ-on-Chip Initiative (NOCI) so valuable throughout my PhD project: it functions as intersection between engineers and biologists and helps us to understand and integrate each other’s disciplines to create in vitro model systems with increased physiological relevance. During the many retreats and practical trainings, organized for and by the NOCI PhD students and Post Docs, we have not only developed our knowledge in Organ-on-Chip technology, but also created a close community, which I am sure will stay connected long after our PhD and Post Doc projects have finished.
With a couple of months still to go, I can already look back on a very rewarding and stimulating PhD that will form a solid basis for an exciting career!

Renée Moerkens
NOCI PhD-student at UMCG
